Summary
Labeling for IV fluids, blood bags, and other liquid-filled medical components presents an elevated risk for contract development and manufacturing organizations (CDMOs). Flexible containers, cold-chain storage, and high-mix production require labels that remain legible and accurate throughout filling, storage, and handling. FDA data show that labeling-related issues are frequent contributors to medical device and drug recalls, underscoring the importance of controlling the labeling process. This article examines how in-house digital labeling — supported by integrated hardware, materials, software, and service from AstroNova Product Identification — helps CDMOs improve traceability, reduce changeover friction, and maintain compliance in sterile liquid manufacturing.
Why labeling is uniquely challenging for IV bags and blood products
Flexible IV bags and blood bags present labeling challenges that rigid containers do not. As bags are filled, they change shape. During storage and transport, they may be exposed to refrigeration, freezing, moisture, and repeated handling. Labels must conform to these surfaces while remaining readable throughout the product lifecycle.
Moreover, governing regulations require labels to carry dense, variable information such as lot numbers, expiration dates, and traceability codes. Errors or unreadable labels are treated as safety risks rather than as merely cosmetic defects. FDA data show that labeling-related issues are a frequent contributor to medical device and drug recalls, underscoring the importance of legibility and accuracy.
High-mix CDMO environments increase labeling risk
CDMOs face a different set of challenges than single-brand manufacturers. Production routinely shifts between clients, formulations, and volumes. Label requirements can vary by product, destination, or customer specification — making pre-printed labels difficult to manage. The challenges include:
- Frequent changeovers between client branding
- Variable data requirements by batch or formulation
- Fluctuating demand that makes label inventory unpredictable
- Increased risk of mismatches between product and label
For liquid-filled medical components, these pressures make labeling for IV bags and blood bags especially complex in high-mix production environments.
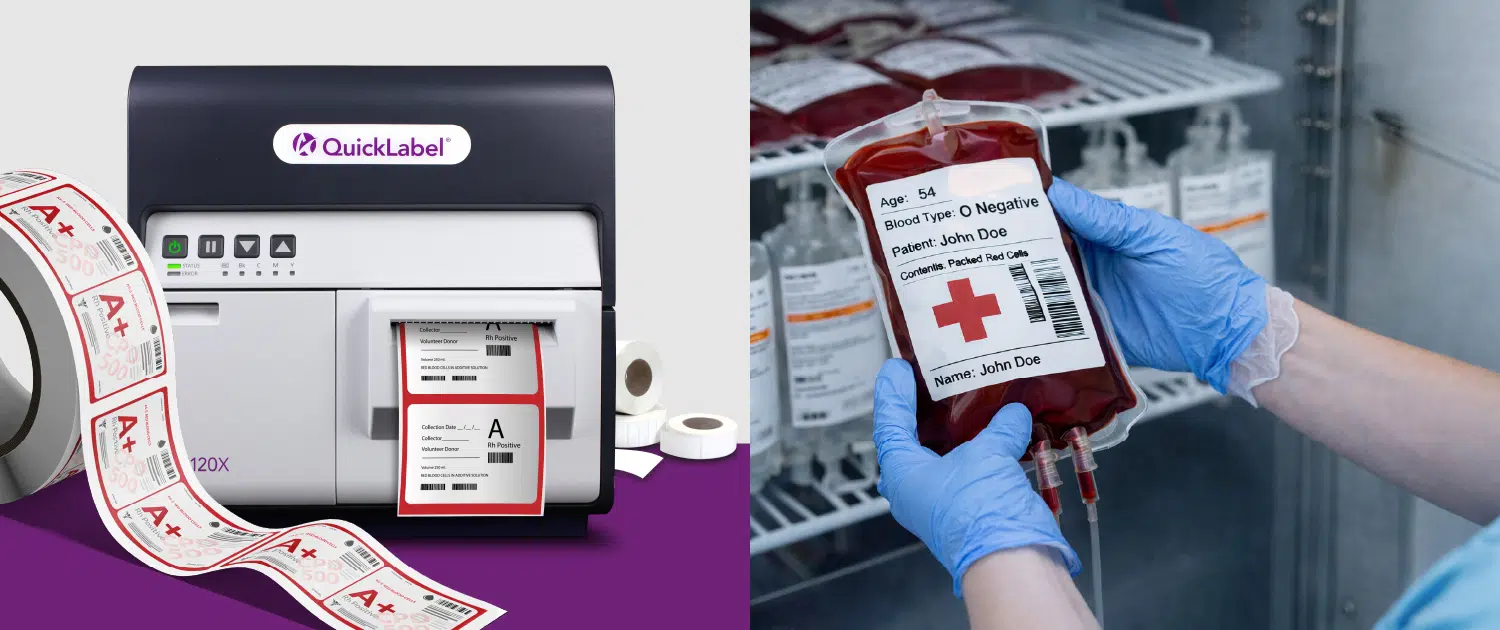
In-house digital label printing as a control strategy
For high-mix sterile operations, in-house digital label printing for medical manufacturing enables CDMOs to produce compliant labels on demand, after product data is finalized. This approach reduces dependency on pre-printed inventory and allows rapid response to specification changes.
Color label printers such as the QuickLabel QL-120Xe and the QL-300 (CMYK+White) support high-resolution output suitable for small label formats and dense information. Clear text and scannable barcodes help ensure traceability, while color can be used to draw attention to warnings and notices without sacrificing readability.
Because templates can be updated immediately, digital printing supports compliance without delaying production.
Label materials must match sterile environments
Printing technology alone does not ensure label performance. Label material selection is as critical as print quality for liquid-filled medical components. For IV bags and blood bags, label materials and adhesives must be selected to withstand real-world conditions encountered during shipment, storage, and use.
AstroNova provides blank, die-cut label materials engineered to support the labeling needs of CDMOs. Synthetic label materials such as polypropylene (BOPP) and polyester are commonly used for IV bags, blood bags, and other flexible medical containers because they conform to curved surfaces and resist moisture and condensation. Permanent acrylic adhesives are typically used in conjunction to reduce edge lift during cold storage and repeated handling.
For CDMOs, AstroNova helps ensure — without the added complexity and cost of sourcing materials and supplies from multiple vendors:
- Correct label formats for IV bags, blood bags, and small medical components
- Adhesives suitable for cold storage, moisture exposure, and handling
- Consistent material availability to help avoid production delays
Software-driven accuracy and traceability
For CDMOs, AstroNova’s CQL Pro advanced labeling software supports consistent labeling through standardized templates and structured variable data fields, including variable data printing for lot numbers, expiration dates, and barcodes. By reducing reliance on manual data entry, the software helps ensure the correct data appears on each product.
In regulated manufacturing environments, CQL Pro reinforces traceability and audit readiness by standardizing data formats across clients, batches, and production lines.
Integrated service for scalable CDMO operations
An integrated service platform for labeling operations helps CDMOs manage complex workflows, maintain uninterrupted production, and support scalability. AstroNova’s service offerings include:
- Technical support for printers and labeling workflows
- Application guidance for label materials and adhesives
- Teams familiar with compliance-driven manufacturing operations
Conclusion
Labeling for IV fluids, blood bags, and life-support components demands durability, accuracy, and operational agility. By aligning printing hardware, label materials, software, and support, CDMOs operating in high-mix regulated manufacturing environments can reduce labeling risk while supporting compliant, scalable production of liquid and life-support medical products.




